I’ve pulled failed seals from acid service where the elastomer looked perfect — but the SiC face was eaten through because it was reaction bonded, not sintered. A seal is only as resistant as its weakest component, and most compatibility resources only cover one component at a time. The charts below cover all three — face materials, elastomers, and metals — organized by chemical family, with temperature and concentration caveats that lab-condition charts leave out.
How to Read This Chart
Each material-chemical pair is rated A through X:
- A — Recommended. No significant attack under normal operating conditions.
- B — Acceptable with limitations. Check temperature and concentration notes.
- C — Marginal. Short-term or low-concentration exposure only. Confirm with testing.
- X — Not recommended. Rapid degradation, swelling, or dissolution expected.
Ratings assume ambient temperature (roughly 70 °F / 21 °C), moderate concentrations, and static immersion. Real-world seals run hotter, under pressure, and with dynamic flexing — all three shift these ratings downward.
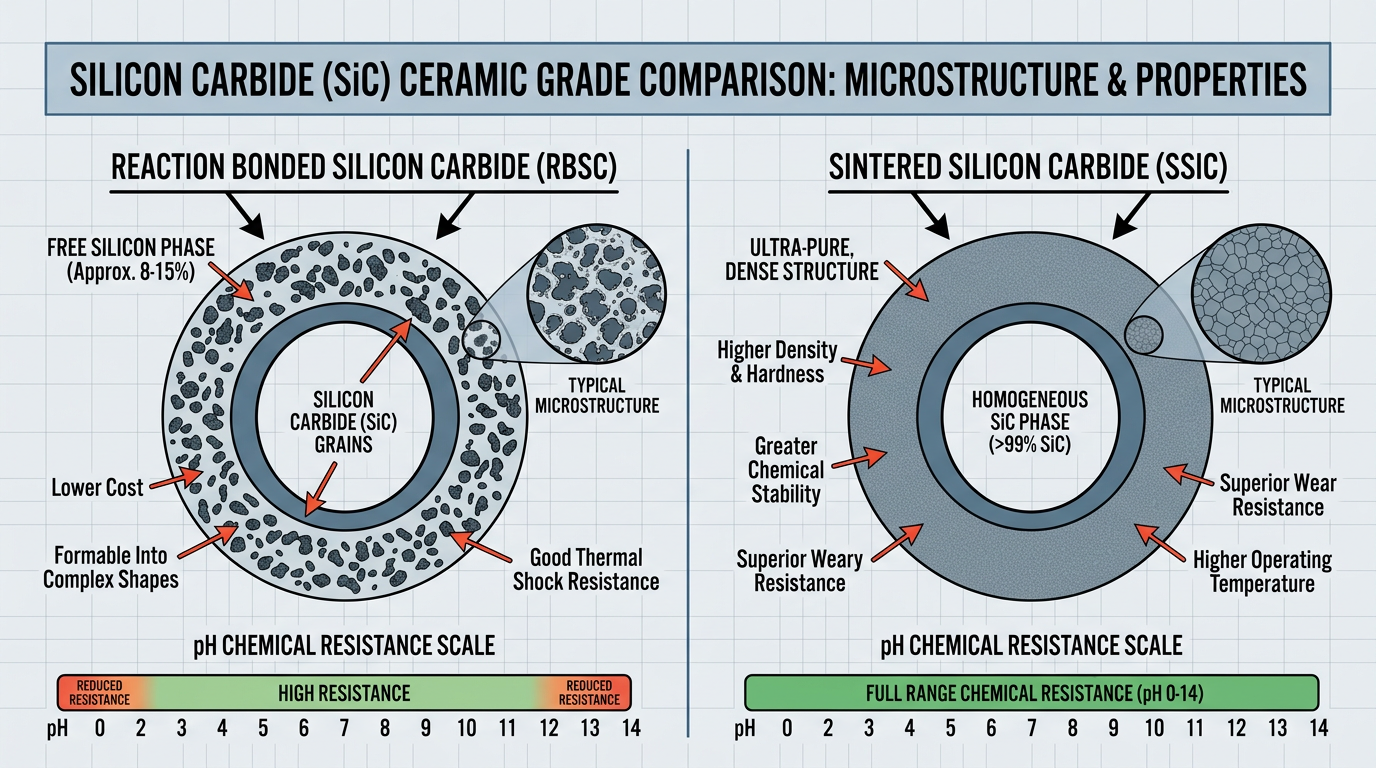
One critical distinction before you look at the face material chart: “SiC” is not one material. Reaction bonded SiC (RB-SiC) contains 8-12% free silicon and must be avoided below pH 4 or above pH 11. Direct sintered SiC (SSiC) is pure silicon carbide with no pH restriction. Most charts list “SiC” without telling you which grade. That omission has caused more field failures than I can count.
Seal Face Material Compatibility by Chemical Family
Resin-filled carbon (#9) tops out at 500 °F (260 °C) and carries FDA approval. Antimony-filled carbon (#10) — the API 682 standard — handles 700 °F (370 °C) with blister resistance that makes it the better choice anywhere gas permeation is a concern. The API 682 standard specifies #10 carbon for a reason — 200 °F of extra headroom plus resistance to subsurface blistering under thermal cycling.
| Chemical Family | Carbon #9 | Carbon #10 | RB-SiC | SSiC | Tungsten Carbide | Ceramic (Al₂O₃) |
|---|---|---|---|---|---|---|
| Mineral acids (H₂SO₄, HCl, HNO₃) | B | A | X (| A | A | B | |
| Organic acids (acetic, citric, lactic) | A | A | A | A | A | A |
| Strong caustics (NaOH, KOH >10%) | B | B | X (>pH 11) | A | A | B |
| Mild caustics (NaOH <10%) | A | A | B | A | A | A |
| Hydrocarbons (gasoline, diesel, crude) | A | A | A | A | A | A |
| Aromatic solvents (toluene, xylene) | A | A | A | A | A | B |
| Chlorinated solvents (methylene chloride) | B | A | A | A | A | C |
| Ketones and esters (acetone, MEK) | A | A | A | A | A | B |
| Ammonia and amines | B | B | A | A | A | B |
| Oxidizing agents (H₂O₂, bleach) | C | B | A | A | B | B |
| Water and steam | A | A | A | A | A | A |
| Food and beverage (CIP solutions) | A | A | C | A | A | C |
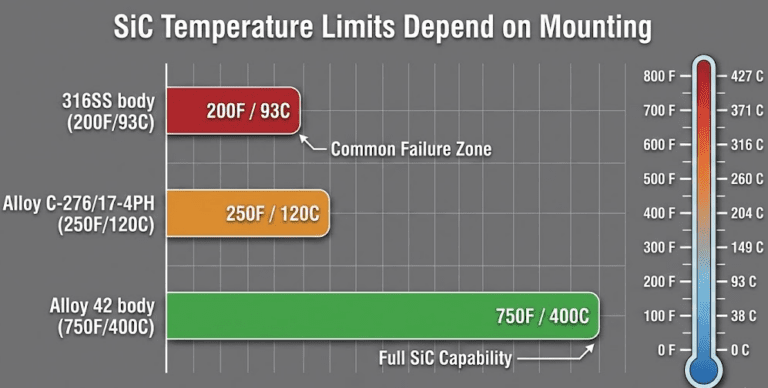
One temperature caveat most engineers miss: a SiC ring pressed into a 316SS holder is limited to 200 °F (93 °C) — not because of the SiC, but because of differential thermal expansion cracking the insert. The same SiC ring in an Alloy 42 holder handles 750 °F (400 °C). The face material rating alone does not tell the full story.
For CIP/SIP processes, ceramic’s brittleness under thermal shock makes it a liability. I recommend SSiC against SSiC for any application involving rapid temperature cycling — the chemical resistance and thermal shock tolerance of sintered SiC justify the cost premium.
Elastomer and Secondary Seal Compatibility
FKM (Viton) covers the broadest range of industrial chemicals at temperatures from 0 °F to 400 °F (–18 °C to 204 °C). EPDM handles what FKM cannot — strong caustics and steam — but falls apart in hydrocarbons. FFKM (perfluoroelastomer, such as Kalrez) resists nearly everything from –4 °F to 620 °F (–20 °C to 327 °C), but at many times the cost of FKM.
| Chemical Family | FKM | EPDM | FFKM | PTFE |
|---|---|---|---|---|
| Mineral acids (<80% conc.) | A | C | A | A |
| Mineral acids (>80% conc.) | B | X | A | A |
| Organic acids | A | B | A | A |
| Strong caustics (>10%) | X | A | A | A |
| Mild caustics (<10%) | B | A | A | A |
| Hydrocarbons | A | X | A | A |
| Aromatic solvents | A | X | A | A |
| Chlorinated solvents | C | X | A | A |
| Ketones and esters | X | A | A | A |
| Ammonia and amines | X | A | A | A |
| Oxidizing agents (H₂O₂, bleach) | C | B | A | A |
| Steam (>250 °F) | X | A | A | A |
| Water (ambient) | A | A | A | A |

I worked with an injection molding facility running caustic chemicals at 400 °F. They burned through standard elastomers every few weeks — swelling, cracking, production shutdowns. Switching to a high-temperature FFKM compound eliminated the failures entirely. The seal cost tripled, but the downtime cost dropped to zero.

Above 350 °F, consider flexible graphite (Grafoil) as a secondary seal element instead of any elastomer. It is chemically inert, handles temperatures that destroy every elastomer except specialty FFKM compounds, and does not suffer from compression set. For high-temperature acid or caustic service, flexible graphite is often the most reliable option.
When elastomers absorb the process chemical, they swell. That swelling increases friction on dynamic O-rings, makes reassembly difficult, and prevents the seal from maintaining proper contact. The damage is not always visible — an elastomer can look intact while its hardness has already dropped enough to lose structural integrity.
Metal Component Compatibility
The wetted metal parts — springs, set screws, retainer rings, and gland plates — see the same chemical environment as the faces and elastomers. 316 stainless steel handles most applications but has clear failure zones.
316SS performs well in organic acids, caustics, hydrocarbons, ammonia, steam, and clean water. It degrades in concentrated mineral acids (rated X), struggles with chloride-containing water (rated C), and shows marginal resistance in oxidizing agents and chlorinated solvents (rated B). Concentrated sulfuric or hydrochloric acid will corrode 316SS springs within months.

Hastelloy C-276 rates A across virtually every chemical family — mineral acids at any concentration, caustics, solvents, chlorides, oxidizers. It is the default upgrade when 316SS cannot survive the environment, but it adds considerable cost to every wetted component.
Alloy 20 sits between the two. It excels specifically in sulfuric acid service (rated A at all concentrations) and handles most other environments well, with slightly lower chloride resistance than Hastelloy (rated B vs A). For sulfuric acid pumps, Alloy 20 delivers Hastelloy-level acid resistance at lower cost.
Specify the metal components with the same care as the seal face materials. A carbon-against-SiC seal with perfect chemical compatibility on the faces and elastomer will still fail if the spring corrodes and loses its closing force.
What Shifts These Ratings in the Field
Every rating in the charts above was established under laboratory conditions — room temperature, single-chemical immersion, no mechanical stress. Field conditions add three variables that shift those ratings, often by one or two grades.
Temperature acceleration. Chemical attack rates roughly double for every 18 °F (10 °C) rise in process temperature. A material rated “A” at ambient can perform like a “C” at 200 °F. Carbon #9, rated to 500 °F mechanically, starts showing accelerated oxidation well below that limit in strongly oxidizing environments. Always cross-reference the temperature limit with the specific chemical, not just the material’s general rating.
Pressure forcing. Higher seal chamber pressure forces chemicals deeper into elastomer and carbon structures. Surface-level compatibility testing does not capture this — the material can pass a static soak test while failing under 150 psi of differential pressure in service. Subsurface degradation is invisible until the seal face delaminates or the O-ring extrudes.
Dynamic exposure. A rotating seal face continuously exposes fresh material to chemical attack, unlike a static gasket sitting in the same fluid. Dynamic flexing of elastomer secondary seals opens micro-cracks that accelerate chemical penetration. This is why corrosive media material selection must account for duty cycle, not just fluid identity.
Process fluid changes create another trap. Facilities switching hydraulic fluids for cost savings or environmental compliance often overlook the seal materials qualified for the original fluid. The new fluid may be compatible with the elastomer but attack the face material, or vice versa. Every fluid change should trigger a compatibility recheck across all three components.
Putting It Together
The fastest path to a correct material selection: identify your chemical family, check all three columns (face, elastomer, metal), then apply temperature and concentration derating. A “B” rating at ambient with a process temperature of 250 °F should be treated as a “C” until validated.
For aggressive or mixed-chemical environments, SSiC faces paired with FFKM secondary seals and Hastelloy C-276 metal parts cover the widest chemical range — but at a cost that only makes sense when the process demands it. Start with the charts, then verify against your actual operating temperature, concentration, and pressure. The chart is a starting point, not a specification.